|
The energy associated with the quantum mechanical change primarily determines the frequency of the absorption line but the frequency can be shifted by several types of interactions. rotation–vibration transitions), leading to new absorption lines at the combined energy of the two changes. X-ray absorptions are associated with the excitation of inner shell electrons in atoms. Electronic lines correspond to a change in the electronic state of an atom or molecule and are typically found in the visible and ultraviolet region. Vibrational lines correspond to changes in the vibrational state of the molecule and are typically found in the infrared region. Rotational lines are typically found in the microwave spectral region. Rotational lines, for instance, occur when the rotational state of a molecule is changed. 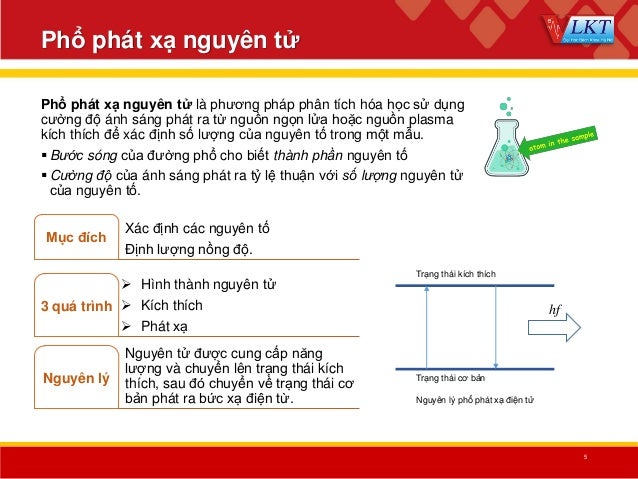
The lines will also have a width and shape that are primarily determined by the spectral density or the density of states of the system.Ībsorption lines are typically classified by the nature of the quantum mechanical change induced in the molecule or atom. The frequencies will also depend on the interactions between molecules in the sample, the crystal structure in solids, and on several environmental factors (e.g., temperature, pressure, electric field, magnetic field). The frequencies at which absorption lines occur, as well as their relative intensities, primarily depend on the electronic and molecular structure of the sample. The absorption that occurs due to a transition between two states is referred to as an absorption line and a spectrum is typically composed of many lines. Radiation is more likely to be absorbed at frequencies that match the energy difference between two quantum mechanical states of the molecules. The absorption spectrum is primarily determined by the atomic and molecular composition of the material. Solar spectrum with Fraunhofer lines as it appears visuallyĪ material's absorption spectrum is the fraction of incident radiation absorbed by the material over a range of frequencies of electromagnetic radiation. The source, sample arrangement and detection technique vary significantly depending on the frequency range and the purpose of the experiment.įollowing are the major types of absorption spectroscopy: Sr. The transmitted energy can be used to calculate the absorption. The most common arrangement is to direct a generated beam of radiation at a sample and detect the intensity of the radiation that passes through it. There is a wide range of experimental approaches for measuring absorption spectra. Absorption spectroscopy is also employed in studies of molecular and atomic physics, astronomical spectroscopy and remote sensing. Infrared and ultraviolet–visible spectroscopy are particularly common in analytical applications. Absorption spectroscopy is performed across the electromagnetic spectrum.Ībsorption spectroscopy is employed as an analytical chemistry tool to determine the presence of a particular substance in a sample and, in many cases, to quantify the amount of the substance present. 
The intensity of the absorption varies as a function of frequency, and this variation is the absorption spectrum. The sample absorbs energy, i.e., photons, from the radiating field. 
Sodium in the atmosphere filters the starlight of HD 209458 as the giant planet passes in front of the star.Ībsorption spectroscopy refers to spectroscopic techniques that measure the absorption of electromagnetic radiation, as a function of frequency or wavelength, due to its interaction with a sample. When exploding fireworks, you will see the color when the metal atoms absorb energy from the detonater.The first direct detection and chemical analysis of the atmosphere of an exoplanet, in 2001.(CC BY-NC-SA 3.0 Christopher Auyeung via CK-12 Foundation)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
